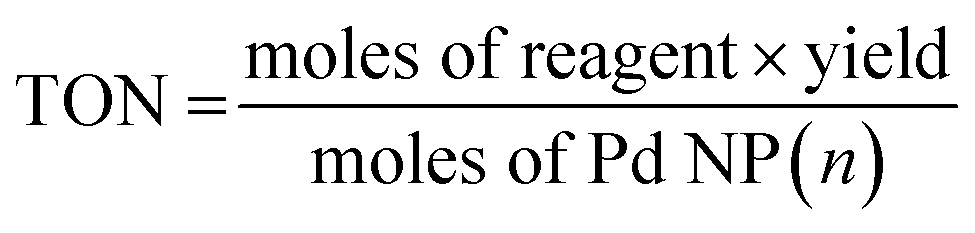
Catalytic farming: reaction rotation extends catalyst performance - Chemical Science (RSC Publishing) DOI:10.1039/C8SC04188A
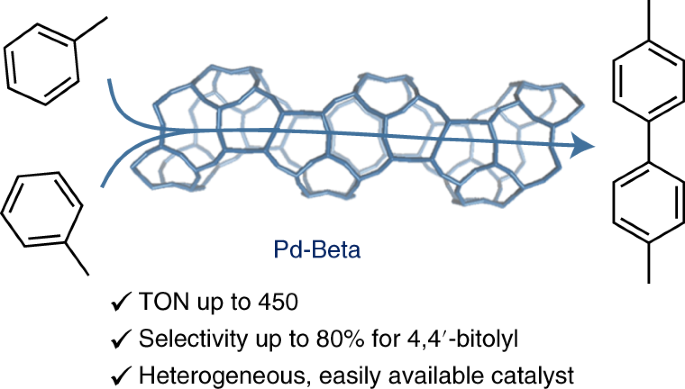
Shape-selective C–H activation of aromatics to biarylic compounds using molecular palladium in zeolites | Nature Catalysis

Molecular ligand modulation of palladium nanocatalysts for highly efficient and robust heterogeneous oxidation of cyclohexenone to phenol | Science Advances

N‐Heterocyclic Imine‐Supported Bimetallic Cu(II) Catalyst for Azide‐Alkyne Cycloaddition: Solvent‐free, Reductant‐free, ppm‐level Catalysis to Access 1,4‐Disubstituted Triazoles - Revathi - Chemistry – An Asian Journal - Wiley Online Library
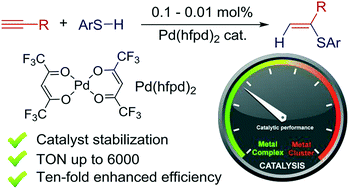
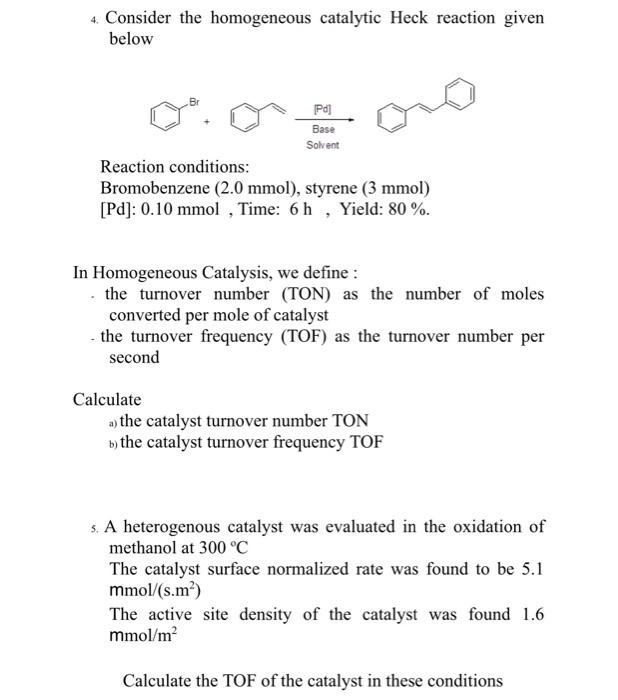
Ten-fold boost of catalytic performance in thiol–yne click reaction enabled by a palladium diketonate complex with a hexafluoroacetylacetonate ligand - Catalysis Science & Technology (RSC Publishing)
How are the turnover number (TON) and turnover frequency (TOF) being used to evaluate and compare different solid heterogeneous catalysts on the same reaction? - Quora
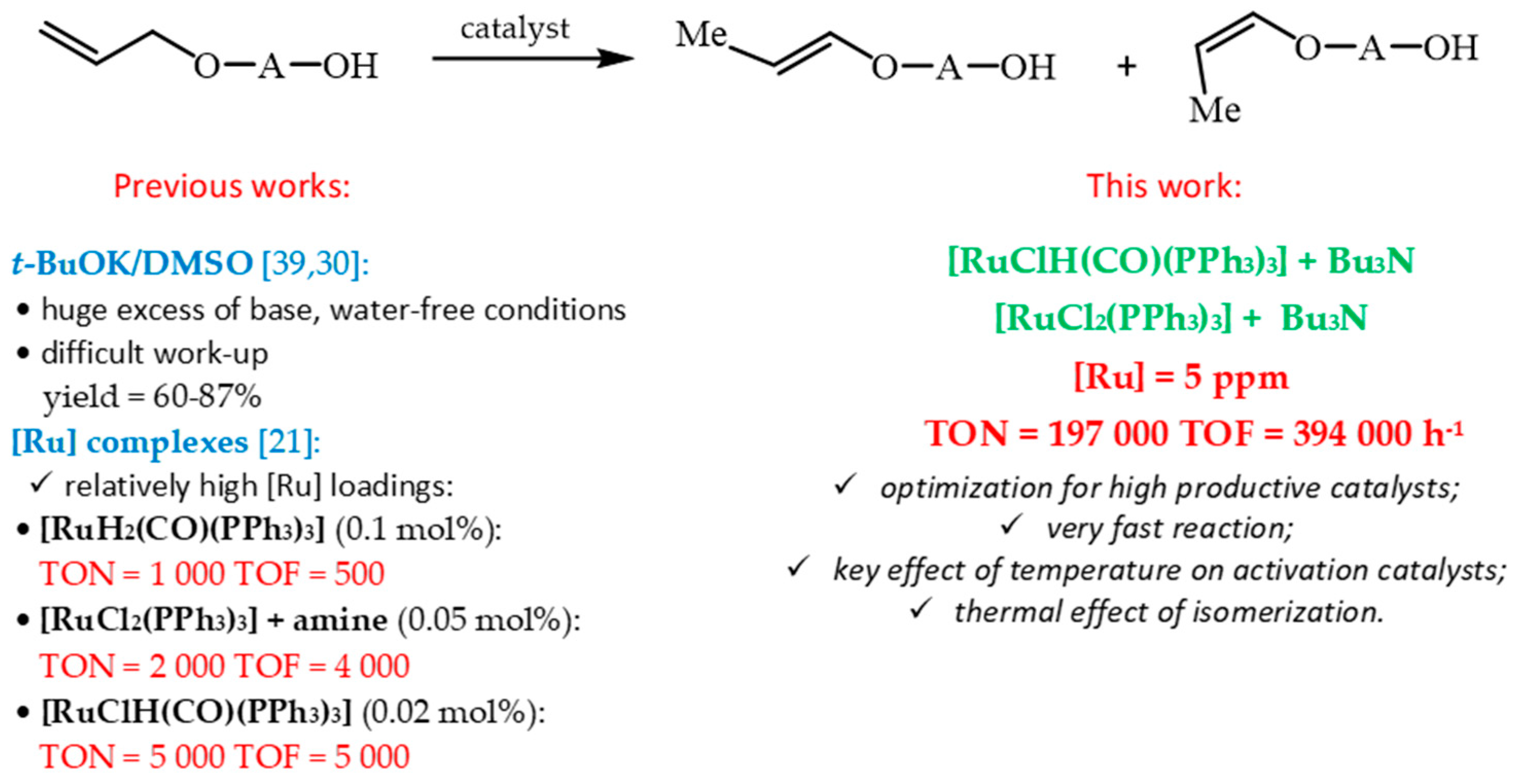
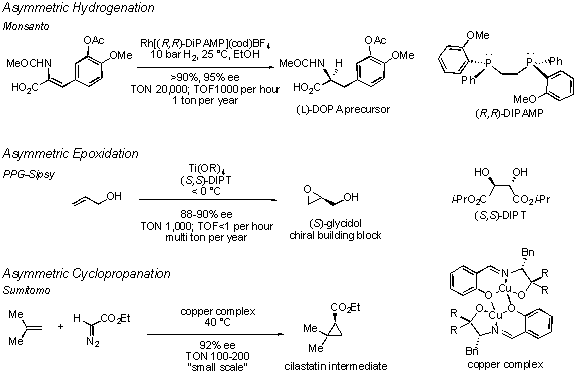
Catalysts | Free Full-Text | Highly Productive Synthesis of 1-Propenyloxybutan-1-ol Under Solvent-Free Homogeneous Ruthenium Catalyst Conditions

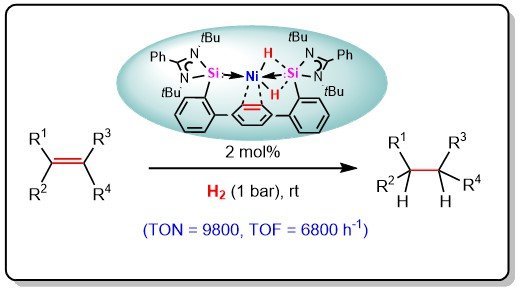
Structure Function Relationships in Ruthenium Carbon Dioxide Reduction Catalysts with CNC Pincers Containing Donor Groups - Das - 2020 - European Journal of Inorganic Chemistry - Wiley Online Library